The Problem
A well-known label manufacturer had a big problem. Their process had changed – managed by an overseas vendor. And now they feared the label or perhaps the label adhesive might contain chemicals in levels that were unacceptable for pharmaceutical applications. Could the solvents used in the label glue present a risk to patients who take the tablets from those containers?

The Issues
The glue was almost impossible to collect from the labels that were submitted to the lab. The adhesive in question was sandwiched between two plastic sheets. Generating adequate samples for testing became extremely difficult.

The Assignment
Recommended as a consultative problem solver with deep experience in leachables and extractables, Quantum Analytics Group was asked to meet with the manufacturer and suggest a plan. Complete testing of all known and unknown solvents used in the manufacturing process was the most immediate hurdle.

The Challenge
Typically, standard methods can be employed when looking for residual solvents in a product (lab receives glue samples) but for reasons unknown…the manufacturer refused to release samples of the actual glue to be tested. By order of the manufacturer, only the finished product (plastic labels) could be tested.
The Timing
The label manufacturer was desperate to respond to their customer…quickly. The timeline associated with the labelling process back in the States could not be changed. Quantum was given just three weeks to test and record the glue samples.

The Solution
The first step was to transfer the clients’ method to Quantum, through a protocol developed by Quantum. The transfer protocol covered detailed expected method performance, criteria for acceptance of data from each laboratory performing the method, and equivalency acceptance criteria between the originating and transferring laboratories. Once successfully transferred, a side by side comparison between the client’s method and the USP method was performed.

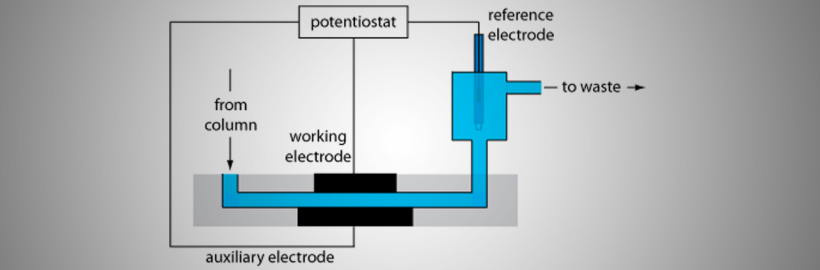
Coulometric detectors can pose challenges not common to other HPLC detectors. Some of the issues that can be encountered include problems with (a) oxidation of some impurity in the mobile phase, (b) deterioration of the working electrode surface, (c) electrode fouling, (d) subtle changes in the temperature of the room or mobile phase, as well as, the slow desorption of electro-active contaminants from the column, tubing or pump heads.

.png?width=100&height=104&name=Frame%204%20(1).png)



